ECO MEDICS
EXHALYZER®Plus
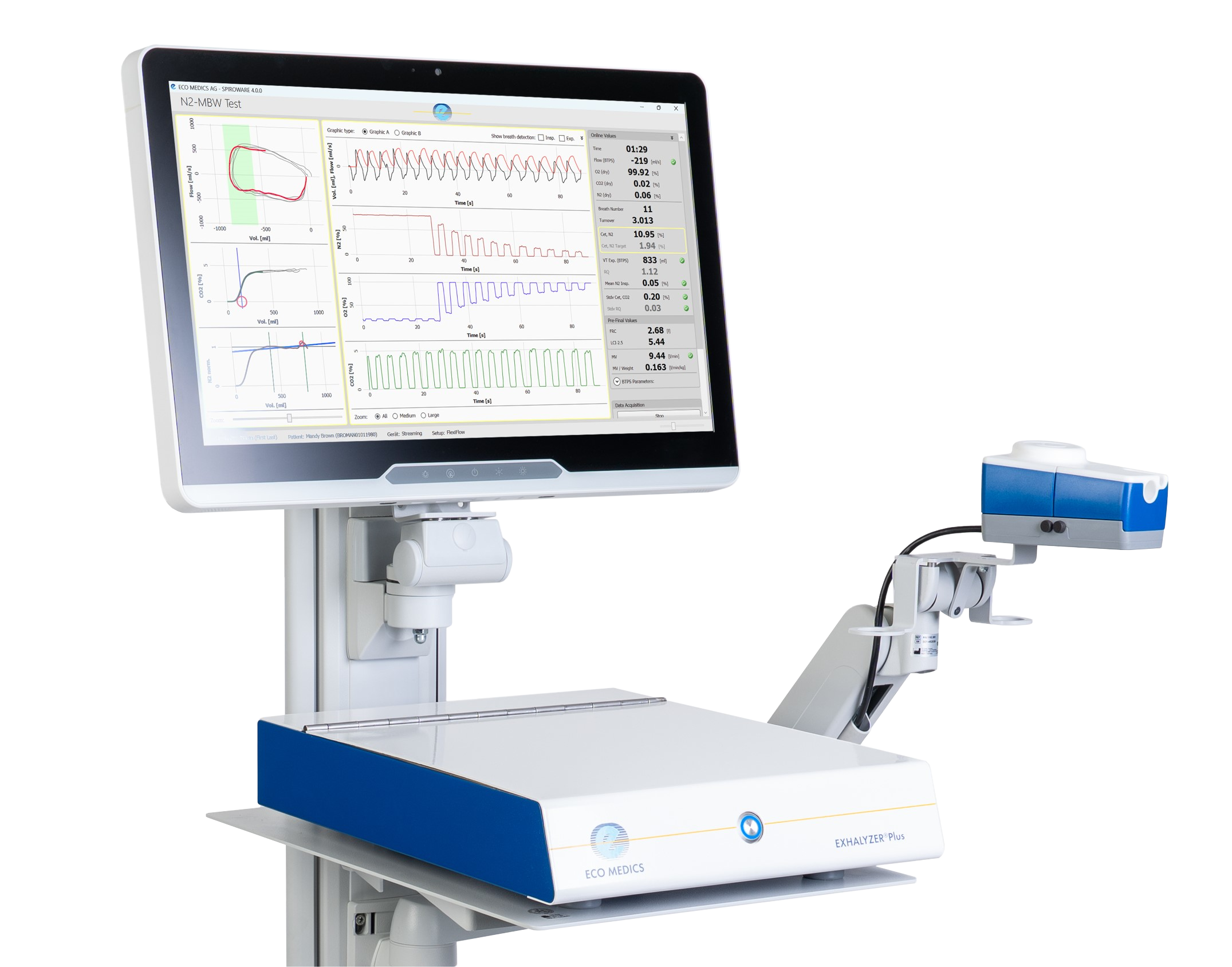
Precision Made Simple
- All-in-one PFT Device for LCI and Spirometry
- Automatic Gas Calibration
- Ergnomic Arm for enhanced ease-of-use
- Optimized SPIROWARE® 4.0 Software
- MDR approved for clinical use
- ATS/ERS Recommendations Compliant
- USA: “For Research Use Only. Not for use in diagnostic procedures.”
ECO MEDICS offers an innovative range of pulmonary diagnostic products for children to adult applications. The focus is the detection of small airway disease by nitrogen washout (N2-washout), FRC / LCI tests and the asthma management as well as PCD screening by FeNO analysis.
FeNO Testing
Exhaled nitric oxide analysis for asthma management and PCD screening in cooperative and non-cooperative patients
ANALYZER CLD 88 sp – FeNO analysis by single and multiple breath testing; bronchial, alveolar and nasal FeNO
Pulmonary Function Testing
Detection and monitoring of ventilation inhomogeneity and small airway diseases such as asthma, cystic fibrosis and COPD
EXHALYZER D – Nitrogen washout by single breath or multiple breath (SBW or MBW), breathing pattern (TBFVL), FRC, LCI, moment ratios and slope analysis
Infant Pulmonary Function Testing
Suitable for cooperative and non-cooperative patients from infants >3kg body weight to adults
EXHALYZER D – Breathing pattern (TBFVL), FRC, LCI and analysis for patients > 3 kg body weight
Liquid NO analysis
Nitric oxide analyzer for liquid samples from biomedical or pharmaceutical applications
ANALYZER CLD 88 – Liquid NO detection in combination with liquid purge vessel and chromatographic software
Sophisticated solutions for our customers
Worldwide distribution.
We acquire the most suitable partners – internally and externally – to develop and manufacture our products according to highest standards and to offer our customers superior service.
ECO MEDICS applies a process oriented management system, which is subject to continuous improvement. Products are CE MDD approved in accordance to ISO 13485 and the Council Directive 93/42/EEC for clinical use.
Limited use by Federal law in the Unites States (non-FDA approved device) and Canada
For Research Use Only. Not for use in diagnostic procedures.
Events
See our products at these upcoming events